physiological processes | stallions | physiological processes
Mares
Introduction:
Successful pregnancy involves not only the mare and stallion, but also the mare’s owner and veterinarian. This is particularly true when using cooled, transported semen where the responsibility of reproductive management of the mare falls directly on the mare's owner and veterinarian. It is more important now than ever for mare owners to understand the physiologic processes involved in the estrus cycle and pregnancy. This discussion will help the mare owner obtain a basic understanding of the following terms regarding the reproductive processes in mares:|
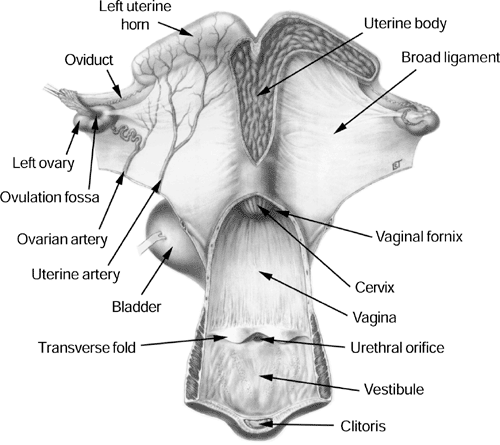
Anatomy Pineal Gland Hypothalamus Pituitary Gland Ovary Oviduct Uterus Endometrial cups Chorionic Girdle Placenta |
Endocrine Factors Melatonin GNRH FSH LH Estrogen Progesterone Oxytocin |
Physiological Processes Estrous Cycle Estrus Diestrus Anestrus Ovulation Capacitation Fertilization Placentation |

Used with permission from OJ Ginther: Reproductive Biology of the Mare: Basic and Applied Aspects, Edition 2, 1992, Equiservices, Cross Plains WI
Physiological Processes:
Mares will cycle during the months of spring, summer, and fall. During the
winter, most mares will become anestrus and stop cycling. The seasonality
of reproductive function is primarily controlled by the pineal gland. The
pineal gland is located deep within the brain and has nerve connections to the
eyes. During the nighttime hours, the pineal gland releases a hormone called melatonin.
Melatonin in turn acts on cells within the hypothalamic region of the
brain to inhibit (limit) production of another hormone, gonadotropin
releasing hormone (GnRH). Therefore, during the spring, summer and fall,
when the nights are shorter, there is less inhibition of GnRH by the pineal
gland.
Nerve tracts carry GnRH from the hypothalamus toward the pituitary gland where it is released into the bloodstream and carried directly through the tissues of the anterior pituitary gland. Certain cells within the anterior pituitary produce either follicle stimulating hormone (FSH) or luteinizing hormone (LH) depending on whether progesterone or estrogen is of dominant influence. The estrous cycle can be divided into two phases, the receptive phase (estrus) and the non-receptive phase (diestrus). If the mare is in diestrus, progesterone is dominant and FSH is released. On the other hand, LH is released under estrogen dominance when the mare is in estrus.
The hormones FSH and LH are called gonadotropins because they are responsible for stimulating activity of certain structures in the mare's ovaries (gonads). FSH activates cells that surround oocytes that have been dormant since even before the mare was born. Only a few oocytes are activated at a time so that the number of oocytes present at birth will be sufficient to last for her entire life. As an individual oocyte is recruited, fluid begins to build up around the oocyte and a follicle is formed. Among the group of follicles that grow each cycle, usually only one becomes dominant. As the dominant follicle grows, estrogen secretion increases and the mare responds by coming into estrus.
Aside from the effects on LH production, estrogen plays an essential role in preparing the reproductive tract for mating. The vestibular vault and the cervix, which are normally coated with a sticky mucus designed to keep contaminants out, during estrus become coated with a thinner, slippery mucus. During estrus the uterus functions as a passageway for sperm cells to enter the oviduct where they can be safely stored for a period of a few days. After copulation, uterine contractions help transport sperm to the oviduct. Sperm are allowed about 2-3 hours for entry; then any sperm remaining in the uterus are actively expelled through the cervix.
Sperm that have entered the oviduct become attached to the cells lining the walls of the oviduct and undergo a process called capacitation. Some cells become capacitated in a few minutes, while others may take days. Regardless of how long it takes, once capacitated, sperm detach from the oviductal walls. They then swim up the oviduct toward the fertilization site. Meanwhile, in the ovary, the follicle containing the oocyte continues to grow and circulating estrogen and LH levels continue to increase. By stimulating LH, follicular estrogen literally signals its own demise since LH will cause ovulation and promote formation of a corpus luteum (CL) in place of the burst follicle.
Upon ovulation, the oocyte is released into the oviduct and shuttled toward the sperm cells that have undergone capacitation and are waiting at the fertilization site. Once fertilized, the newly formed embryo will be nurtured in the oviduct for approximately 5-6 days. Then, through a mechanism that is not completely understood, the embryo will be pushed out into the uterus where it will continue to grow.
By the time the embryo enters the uterus, the CL is mature and the progesterone released by the CL has physiologically transformed the uterus to provide a supportive environment for the developing embryo. In contrast to many other mammals, the embryo of the horse undergoes a prolonged mobility phase until about 16 to 17 days after ovulation. Afterwards it settles at the lowest point of either horn located near the bifurcation. During this mobile phase, the embryo signals the uterus to prevent prostaglandin release and subsequent lysis (destruction) of the CL that would otherwise occur at day 14. This is why nonpregnant mares normally return to estrus by day 16.
Cells within the embryo then differentiate (change) into the varied tissues that make up the placenta and fetus between days 18 and 35. On about day 35, certain embryonic cells called chorionic girdle cells migrate from the embryo and penetrate the uterine lining. There they multiply and make identifiable structures called endometrial cups that release a hormone called equine chorionic gonadotropin (ECG) between days 35 and 100 of pregnancy. This hormone has both FSH and LH-like effects on the ovaries since it seems to stimulate both follicular development and the formation of additional, secondary CLs. These CLs are responsible for producing progesterone that helps maintain the pregnancy as the placenta gradually becomes integrally attached to the uterine lining. After day 100, the endometrial cups slough away and the placental tissues become the primary progesterone source supporting the pregnancy to term.
Summary: The establishment of pregnancy in mares requires that a complex series of biological interactions take place. Seasonal and cyclic physiological changes are driven by hormonal influences to support follicular development, ovulation, fertilization, and pregnancy. Understanding these changes is an important step toward acquiring the ability to take an active role in breeding management.
Introduction:
This section will focus on the basic physiological processes involved in sperm production, sperm maturation, and the emission of seminal components. The anatomical structures involved in the hormonal control of sperm production include the pineal gland, hypothalamus, pituitary gland, and testes (testicles). The hormones involved in spermatogenesis (development of sperm) include melatonin, gonadotropin releasing hormone (GnRH), follicle stimulating hormone (FSH), luteinizing hormone (LH), testosterone, androgen binding protein, activin, and inhibin. Within the testis, spermatogenesis involves the action of Leydig cells within the interstitium, and Sertoli cells lining the inside of the seminiferous tubules. The epididymis, ductus deferens, and ampulla are involved in maturation and storage of sperm. Emission of semen involves the tail of the epididymis, ductus deferens, and the ampulla. Seminal fluids are produced by the seminiferous tubules, ampulla, vesicular glands, prostate gland, and bulbourethral gland. Emission of semen into the reproductive tract of the mare is achieved through the urethra during ejaculation. Physiological
Processes:
Just as mares experience seasonality of their reproductive processes,
stallions produce more sperm cells in the spring and summer months. This is
primarily due to reduced inhibition of hypothalamic GnRH by pineal
melatonin during the longer days of the year. Higher GnRH levels in turn
stimulate the release of FSH and LH from the pituitary gland.
These two hormones stimulate activity of certain cell types within the tissues
of the testes.
The testes are made up of several anatomical structures, tissues, and cell types. The structures can be categorized into two basic types, interstitial and tubular. The interstitial region contains cells that provide structure, blood supply, lymph flow, and testosterone production. The cells responsible for testosterone production are called Leydig cells. High levels of testosterone are needed within the testes to support sperm development. Elevated blood testosterone concentrations signal the hypothalamus and pituitary gland to stop secreting GNRH and LH. Likewise, low blood testosterone signals increased GNRH and LH secretion.
Tubular structures called seminiferous tubules lie throughout the interstitial tissues and are directly involved in sperm production, maturation, and transport. Primordial spermatogonia (undeveloped male germ cells) line the outer border within the seminiferous tubules. These cells continually divide to produce both replacement primordial spermatogonia and differentiated spermatogonia. Over the next two months, each differentiated spermatogonia undergo a series of divisions and maturation processes resulting in the potential release of 64 tailed but immobile spermatozoon into the opening (lumen) of the seminiferous tubule.
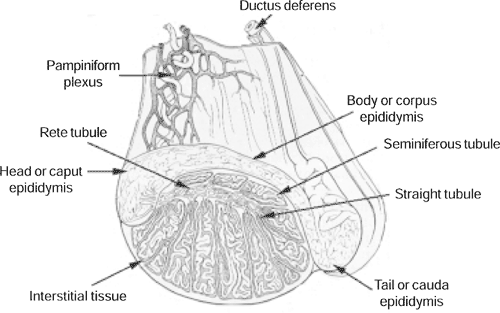
Stallion testis: modified from Pickett, BW, et al.: Management of the Stallion for Maximum reproductive Efficiency.
Recruitment of spermatogonia and their initial developmental processes are controlled by Sertoli cells. Sertoli cells are anchored to the outer border of the tubule and extend to an open lumen in a spoke-like pattern. Sperm cells at varied stages of development can be found at different locations between the outer border and the center of the tubule and between the larger Sertoli cells. The spermatogenic activity of Sertoli cells is supported by FSH from the pituitary gland and testosterone from interstitial Leydig cells. To maintain the proper FSH concentration, they release activin to signal that FSH levels are high and the hormone inhibin to signal the pituitary to stop releasing FSH. Testosterone, like all steroids, can pass freely across the cellular barriers and into the Sertoli cells. However, to ensure that a high concentration of testosterone is maintained within the seminiferous tubule, Sertoli cells produce a specialized molecule called androgen binding protein (ABP) that traps testosterone within the tubule and ensures it is available when needed.
Androgenic steroids have become a popular training aid to enhance performance for some competitive events. Androgens such as Equipoise
TM, immediately inhibit pituitary LH and testicular testosterone production. When local testosterone availability decreases, sperm production suffers. Mature stallions can recover 2-3 months after drug withdrawal. However, in sexually immature stallions (less than 6 years of age), permanent damage may result to the stallions capacity to produce sperm. The extent of this damage will depend on the age of the stallion and duration of androgen exposure. Androgenic steroid administration is especially damaging if given to stallions 3 years of age or less.As spermatids mature, they are released and constant contractions move the immobile spermatozoon through a series of ducts to the head or caput epididymis. These ducts do not generate sperm, rather are involved in absorption of fluid, thereby concentrating the sperm. Next, sperm gradually pass through the body or corpus epididymis. In this region, sperm cells mature and acquire the ability to swim. By the time the sperm reach the tail or cauda epididymis, approximately 2 months has passed from the time the whole process began. Now the sperm cells are fully mature and capable of fertilization.
Upon ejaculation, muscle contractions bring sperm from the paired ductus deferens to be mixed with seminal fluid from the prostate gland, vesicular glands, and bulbourethral glands. The contribution of seminal fluid produced by each of these glands to fertility is unclear since research has shown that sperm cells extracted from the tail of the epididymis have normal fertility. The ejaculate can be divided into 3 parts: the pre-ejaculatory fluid, the sperm rich fraction, and the gel fraction. Pre-ejaculatory fluid is a watery fluid released from the prostate and bulbourethral glands and serves to rinse the urethra. Occasionally, this can be seen dripping from the stallions penis during teasing. The next fraction adds fluid secretion from the ampulla and sperm to the mix. Finally, near the end of the ejaculate, the vesicular glands contract and release a gelatinous substance. When preparing semen for artificial insemination (A.I.), this "gel" fraction is filtered off prior to diluting semen with extender.
It is important that the testicular temperature is maintained approximately 5
o C below body temperature. A number of anatomic mechanisms are in place to regulate testicular temperature. These mechanisms can be defeated by fever, restriction of blood flow, physical trauma, or infection. Reduction or interference in spermatogenesis may result. Due to the duration of spermatogenesis, resumption of normal sperm production and quality will be delayed approximately 2 months from the time the testicular temperature is corrected.Summary:
The reproductive tract of the stallion can most simply be described as a continuous production and delivery system for sperm. The rate of sperm production is subject to seasonal, hormonal, and environmental influences. Understanding these influences may help managers understand the importance of adhering to proper management practices.Selected References:
•Blanchard TL, Varner DD, Schumacher J: Manual of Equine Reproduction,
1998, Mosby Inc.
•Ginther OJ: Reproductive Biology of the Mare: Basic and Applied Aspects,
1993, Equiservices.
•McKinnon AO and Voss JL: Equine Reproduction, 1993, Williams and
Wilkins.
•American Society of Andrology, Handbook of Andrology, 1995, Allen
Press inc.
•Squires El: Understanding the Stallion, 1995, Blood-Horse Inc.
Future updates to this section of the manual will include information on breeding soundness exams, fertility, estrus cycles, teasing, artificial insemination, pregnancy, etc.